MENINGITIS AND BLOOD POISONING
New ‘Superbug’ Meningitis C
Meningitis C “superbugs” that are impervious to vaccines could pose a
new threat to children, new research has claimed.
Scientists in the UK have identified three strains with heightened
resistance to vaccine-triggered immune responses.
At present there are no reports of emerging vaccine-resistant
meningitis C in the community. But experts fear if the strains get a
foothold they could undermine the vaccine, introduced into the UK in 1999.
Each year more than 2,000 people, mostly children, are affected by
all forms of meningitis in Britain. The disease claims about 300
lives per year and leaves hundreds of other victims with permanent
disabilities.
The authors of the new research, led by Professor Christoph Tang,
from Imperial College London, called for more studies into the
genetic changes behind vaccine resistant meningitis C.
A simple alteration in its DNA is thought to give the bug the
potential to avoid attack by immune system antibodies.
The study, published in The Journal of Experimental Medicine, was
funded by the charity Meningitis UK.
Sourced from a UK Press Google article.
VAN UK’S COMMENT: Vaccines are known to mutate and if you suppress an illness it can develop into another form of the illness. Currently the same thing is happening with prevnar causing superbug ear infections.
Meningitis Vaccinations blamed for a rise in deaths
The success of a vaccination campaign against meningitis is being
blamed for a sudden rise in the number of deaths.
Cases of meningitis and septicaemia have fallen from about 4,000 a
year in the late 1990s to 2,446 last year following the introduction
of a vaccine against meningitis C in November 1999. But in a bizarre
twist the number of deaths rose last year by 17 per cent from 317 to
370 and is not far below the level before the vaccine was introduced.
Specialists say one reason for the rise in deaths is the mistaken
belief that the vaccine protects against all forms of meningitis. The
vaccine is only effective against meningitis C, cases of which have
fallen by 90 per cent, but offers no protection against the equally
deadly meningitis B.
The Meningitis Research Foundation is to launch a campaign this week
to alert the public to the risks. A spokes-woman said: “We are
extremely concerned about the rise in deaths. With cases declining
this is the last thing we want to see. We don’t know why deaths are
rising but anecdotal evidence suggests many people think that since
the introduction of the meningitis C vaccine the problem is solved.”
The vaccine is given routinely to all babies in the first months of
life and has also been administered in catch-up exercises to older
children and young adults.
But the foundation says this has generated a false sense of security
among the public and led parents and individuals to ignore symptoms,
leading to delays in getting help.
“In a survey of students we found that half thought that, because they
had been vaccinated, they couldn’t get meningitis. They thought they
were OK and didn’t have to worry any more. Callers to our helpline
said the same thing. So many mums said they didn’t think their child
could get the disease because they had been vaccinated.”
The Independent 20th September 2004.
New military recruit died of meningitis shortly after being vaccinated for it.
A Marine recruit who died Dec. 15 had an overwhelming meningococcal
bacteria infection that was different from the streptococcus A that
infected 185 other recruits at the Marine Corps Recruit Depot, it was
reported Tuesday.
All recruits entering MCRD are vaccinated against the meningococcal
bacteria, but the vaccine is not always effective, Malone said.
One recruit remains in critical condition from the step A-related pneumonia
outbreak that struck the depot.
More than 126 people were hospitalized with pneumonia, though not all were
related to strep A.
http://story.news.yahoo.com/news?tmpl=story&u=/ibsys/20021224/lo_kgtv/1432569
Despite vaccine, meningitis takes teen’s life
When Bentley College freshman Erin M. Ortiz went home sick last weekend,
her mother did what any mother might do. She cooked comfort foods – corned
beef, rice, and plantains – reflecting her daughter’s Puerto Rican and
Irish heritage.
“It was her favorite meal,” said Brenda Rivera, a family friend.
But just hours after complaining of a headache and going to bed to sleep it
off, Ortiz, 18, was dead of bacterial meningitis, a disease against which
she had been vaccinated. Now, Ortiz’s family hopes others will learn from
their story.
“I’m all cried out,” said her father, Raymond Ortiz. “I’ve got a hole in my
heart. I don’t think I’ll ever be the same.”
“We thought she’d be covered,” he said. “They don’t tell you that even if
you get the vaccine, you’re still susceptible.”
By Tania deLuzuriaga, Globe Staff | October 10, 2007 Boston.com news.
IRISH KIDS DEVELOP MENINGITIS DESPITE BEING VACCINATED
Three children have been hospitalised with meningitis in Ireland this year
despite being vaccinated against the disease.
Recommendations were made last July to offer meningitis boosters after it
was found that vaccinations given at two, four and six months may not be
adequate enough to fight the disease caused by the haemophilus influenzae
bacteria (Hib).
However, the booster programme is yet to be implemented in spite of calls
from the National Immunisation Advisory Committee (NIAC).
Consultant paediatrician at Portiuncula Hospital in Ballinasloe, Dr Kevin
Connolly, said: “I am disappointed that the implementation of a booster
programme was not advanced quicker. The NIAC scheduled extra meetings
specifically to discuss whether a booster was necessary.
“We made a decision in July in the hope that a booster dose would be
introduced before the winter period. I am very disappointed that nothing
definite has been decided thus far.”
It also emerged this week that several children have suffered neurological
damage in the past few years as a result of the disease, which can also
cause septic arthritis and a fatal voice box infection.
Despite the development of a new vaccine in 1992, 14 cases of the disease
were reported in 2003, the highest number in almost ten years.
Netdoctor.co.uk
VAN UK’S COMMENT: Since the vaccine clearly didn’t work and may have even contributed to the meningitis why would they think giving more ineffective vaccines would help?
Vaccine Adverse Event Reporting System Reports Of Meningitis After Hib Vaccine
VAERS ID 25493
An 18 month old boy was vaccinated with hib vaccine and 160 days later he developed pneumonia and according to diagnostic lab tests he had blood cultures positive for Haemorphilus Influenzae type B.
VAERS ID 25796
2 year old boy developed Hib blood poisoning and meningitis 310 days after hib vaccination.
He had a history of febrile seizures.
VAERS ID 25798
A 2 year old girl was given a hib vaccine and 102 days later she developed hib meningitis and had temporary paralysis of her lower limbs. Her blood culture tested positive for Haemophilus influenzae B.
VAERS ID 27261
A 6 month old baby recieved his first hib vaccination and 22 days later he was hospitalized with hib sepsis and meningitis.
VAERS ID 27274
An 18 month old girl had a hib vaccination and developed meningitis within 12 hours of the shot. Her blood culture was positive for hib.
She had had a urine infection at the time of vaccination but was otherwise healthy.
19 month old child had Hib vaccine and DPT and oral polio. 504 days later he DIED after contracting hib meningitis. His blood culture tested positive for Hib.
His father had stated he had some kind of immune system disorder prior to vaccination.
VAERS ID 27813
A 2 month old baby boy had Hib vaccine DPT and oral polio vaccines.
He developed hib meningitis and sepsis 10 days after vaccination.
VAERS ID 233312
A 4 month old boy was vaccinated with DTaP Hib and pneumonia vaccines.
36 days later he was hospitalized with hib meningitis and pneumonia. His blood culture tested positive for hib.
Recent 2008 VAERS Failure Reports
A 2 month old boy was vaccinated with DTAP HIB IPV PNC and ROTHB5 vaccines on 3rd April 2008.
3 days later he developed pneumococcal meningitis and bacteremia.
He had been born at 33 weeks.
VAERS ID 320337
A 5 month old boy was vaccinated with hib vaccine and 279 days later he developed hib meningitis which was confirmed by a spinal tap.
VAERS ID 324934
A 15 week old boy DIED after having his vaccinations on 15th July 2008.
Information regarding PREVNAR was received from a pediatrician regarding a 15-week-old male patient who experienced Pneumococcal meningitis. At 15 weeks of age, the patient received the first dose on 15-Jul-2008. He also received the first doses of HIB (manufacturer unknown), Poliomyelitis Vaccine Inactivated (manufacturer unknown), DTaP and ROTATEQ (Merck & Co Inc) on 15-Jul-2008. Relevant medical history was not provided. Concomitant therapy included Hepatitis B Vaccine, HIB, ROTATEQ, DTaP and Poliomyelitis Vaccine Inactivated. On 25-Aug-2008, the patient presented with nystagmus, irritability and fever. He was hospitalized and diagnostic tests were performed (see test results), however serotyping was not performed. The blood culture and cerebral spinal fluid culture were positive for Pneumococcus. The patient was diagnosed as having Pneumococcal meningitis drug ineffective on 25-Aug-2008. Treatments were not reported. The patient died on 28-Aug-2008. The physician considered PREVNAR to be ineffective in preventing Pneumococcal meningitis, but stated that she realized that only receiving one dose in the series did not confer immunity to the disease. No further information was available at the time of this report. The cause of death was reported as meningitis Pneumococcal.
VAERS ID 325003
A 2 month old girl was vaccinated with DTAPHE HIB PNC ROTHB5. Unresponsive after vaccinations and developed a fever.
2 days later she was diagnosed with meningitis.
She had been previously healthy with no pre-existing conditions.
The VAERS reports were only an example of the 33 pages of meningitis after hib vaccine reports that I studied and these are only the cases which were notified by doctors in the USA.
Two Babies Got Meningitis….They Had Both Been Vaccinated
Two Calgary children are receiving hospital treatment for serious cases of the infection that can lead to meningitis, health authorities say.
Mersadiz Clark, 15 months, has been hospitalized since Tuesday, hooked up by tubes to machines to keep her alive, her father, Blake Clark said. Doctors have diagnosed his daughter with bacterial meningitis, he said.
“When we brought her in, she was limp like a sandbag, her head kept flopping to the side and her skin was yellow with red blotches all over,” said the 22-year-old father.
The toddler’s playmate, and cousin, Dayon, 4, is also at the children’s hospital, he said.
Schnurr, 22, said her daughter has been diagnosed with bacterial meningitis, and is unsure how she contracted the infection. She was up-to-date on her vaccinations, Schnurr said, adding Dayon is being treated for the same infection and had also been vaccinated.
Source: Calgary Herald, 23 January 2009.MENINGOCOCCAL DEATH RATE INCREASED 150% AFTER MENZB VACCINE 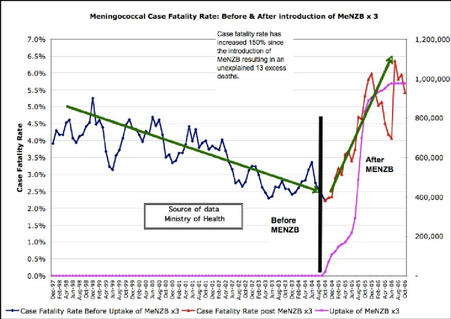
Ministry of Health data reveals the meningococcal disease case fatality rate has increased by 150 percent since the MeNZB vaccine was rolled out, resulting in 13 unexplained deaths.
Norway banned the vaccine because of safety concerns and refused to accept imports of it from France, yet the same vaccine is injected into healthy babies in New Zealand.
Source: Scoop Independent News, 13th November 2006, by Ron Law.
Toddler Dies of Meningitis C, Despite Being Fully Vaccinated
A toddler from Wythenshawe has died from meningitis – despite being vaccinated against the disease.
Lewis Taylor, two, was struck down by meningococcal C – so rare that only a handful of people die from the strain each year. He was rushed to hospital by his parents but doctors could not save him.
Now his devastated parents Darren Taylor, 25, and Rebecca Weedon, 27, have told of their heartache. They want to warn other families of the symptoms to try to stop anyone else going through a similar ordeal.
Lewis, who died on September 12, had been immunised against meningococcal C. He had received three doses of the vaccine – in line with standard immunisation programmes.
Source: Manchester Evening News, September 20, 2011.
Complicated Pneumococcal Meningitis in a Fully Vaccinated Child
We present a unique case of life-threatening pneumococcal meningitis complicated by vasculitis in a fully vaccinated 4-year-old female with the heptavalent conjugate vaccine for Streptococcus pneumoniae (PCV7). Serotype 23F was isolated in both blood and cerebral spinal fluid (CSF) samples. Pulses of methylprednisolone were promptly initiated in addition to antibiotics leading to a good recovery. CSF studies and magnetic resonance imaging (MRI) of the brain were used for diagnosis and monitoring the response to corticoid therapy. We report the investigations of a child with failure of PCV7 vaccine to protect against vaccine-serotype invasive disease. Use of corticosteroids and temporal association with changes in brain imaging are described for the first time in literature.
Source: Neuropediatrics. 2011 Nov 14. http://www.ncbi.nlm.nih.gov/pubmed/22083882
Streptococcus pneumoniae meningitis in a child vaccinated with pneumococcal heptavalent conjugate vaccine
Pneumococcal meningitis is still today a life threatening disease among children under-5 worldwide. Although the heptavalent vaccine has demonstrated its ability to reduce the incidence of pneumococcal disease its efficacy is limited due to the restricted number of serotypes included. We report a case of a child with a Streptococcus pneumoniae meningitis despite the use of heptavalent conjugate vaccine.
Source: New Microbiol. 2009 Jul;32(3):317-8. http://www.ncbi.nlm.nih.gov/pubmed/19845117
Vaccinated 3 Year Old Gets Meningitis C
A DEVASTATED couple were told to “prepare for the worst” after their three-year-old son contracted meningitis C.
Little Euan Couttie developed septicaemia, the blood poisoning form of the disease, and heartbroken mum and dad Siobhan, 32, and Douglas Couttie, 30, were told their son might not survive the night.
The couple have now issued a warning to other parents to be aware of the signs of meningitis after their son almost died, despite receiving the vaccine.
Source: Evening Times, 29th July 2015.
Family’s fears after three-year-old’s meningitis diagnosis
Meningitis B vaccine disappoints in campus outbreak
A study in the New England Journal of Medicine yesterday showed the meningitis B (MenB) vaccine, Bexsero, when administered during an outbreak at Princeton University in 2013, produced an immune response in most recipients, but 34% had no immune response to the outbreak strain.
Researchers began administering the vaccine, made by GlaxoSmithKline, at Princeton in December 2013, when a MenB outbreak began on campus. At the time, the multicomponent meningococcal serogroup B vaccine (4CMenB) was approved for use in Europe and Canada (the Food and Drug Administration approved it in 2015).
All 6,000 Princeton students were offered the vaccine, and 89% of students received at least one dose within 2 months of the outbreak. By the end of the 2-year outbreak, there were 9 cases of MenB reported on campus and 1 death.
“We had the unique opportunity to test the vaccine before licensure in US,” said Nicole Basta, PhD, MPhil, assistant professor of epidemiology and community health at the University of Minnesota. “All students who were tested had an immune response to at least one strain contained in the vaccine, but a third didn’t have any response to the outbreak strain.”
Basta and colleagues looked at 499 students who received two doses of the 4CMenB vaccine over 10 weeks. Two months after receiving the second dose of the vaccine, only 66% of subjects showed bactericidal activity against the MenB strain that was infecting students.
“This level of seropositivity was lower than expected, given the antigenic similarity between the outbreak strain and the components of the vaccine and given that the Meningococcal Antigen Typing System predicted that 4CMenB would induce responses against the outbreak strain,” the authors wrote.
Moving forward
The authors conclude that a third dose of 4CMenB might increase seropositivity during an outbreak. In an editorial on the study, Jerome Kim, MD, director general of the International Vaccine Institute, said that the 4CMenB vaccine could still be an important public health tool.
“For a relatively uncommon but devastating infectious disease, the regulatory approval of a vaccine in the absence of ideal data may be necessary and appropriate if the vaccine is deployed in the context of a systematic public health response,” said Kim.
Though rare, MenB infection can be fatal and can wreak havoc on college campuses. Since 2009, there have been seven outbreaks on college campuses, resulting in 3 deaths and 43 cases. College students are at risk because they live in close quarters, and are known to share food and drink.
Based on the performance of 4CMenB, the Advisory Committee on Immunization Practices, which advises the Centers for Disease Control and Prevention, concluded, “Vaccination of all adolescents would prevent 15 to 29 cases and 5 to 9 deaths annually in the United States.”
Even with those small numbers, Basta said she’d recommend the vaccine to teens and young adults, especially those heading to college.
“Meningitis is still pretty rare in the US, but it can be life-threatening.”
Source: CIDRAP, 21st July 2016.
http://www.cidrap.umn.edu/news-perspective/2016/07/meningitis-b-vaccine-disappoints-campus-outbreak
Immunogenicity of a Meningococcal B Vaccine during a University Outbreak
Background
In December 2013, a multicomponent meningococcal serogroup B (4CMenB) vaccine was used before licensure on the basis of special consideration by the Food and Drug Administration to respond to an outbreak of Neisseria meningitidis B at a U.S. university. Data suggested that vaccination would control the outbreak because isolates expressed antigens that were closely related to the vaccine antigens (factor H–binding protein [fHbp] and neisserial heparin-binding antigen). We quantified the immune responses induced by 4CMenB during the outbreak.
Methods
We conducted a seroprevalence survey among students to assess vaccination status and collect serum specimens to quantify titers of serum bactericidal antibodies (SBA) with an assay that included human complement (hSBA). We compared the proportion of vaccinated and unvaccinated participants who were seropositive for the outbreak strain and for one closely related reference strain (44/76-SL, which included fHbp) and one mismatched reference strain (5/99, which included neisserial adhesin A), both of which were used in vaccine development. Seropositivity was defined as an hSBA titer of 4 or higher.
Results
Among the 499 participants who received two doses of the 4CMenB vaccine 10 weeks apart, 66.1% (95% confidence interval [CI], 61.8 to 70.3) were seropositive for the outbreak strain, although the geometric mean titer was low at 7.6 (95% CI, 6.7 to 8.5). Among a random subgroup of 61 vaccinees who also received two doses but did not have a detectable protective response to the outbreak strain, 86.9% (95% CI, 75.8 to 94.2) were seropositive for the 44/76-SL strain, for which there was a geometric mean titer of 17.4 (95% CI, 13.0 to 23.2), whereas 100% of these vaccinees (95% CI, 94.1 to 100) were seropositive for the 5/99 strain and had a higher geometric mean titer (256.3; 95% CI, 187.3 to 350.7). The response to the outbreak strain was moderately correlated with the response to the 44/76-SL strain (Pearson’s correlation,0.64; P<0.001) but not with the response to the 5/99 strain (Pearson’s correlation,−0.06; P=0.43).
Conclusions
Eight weeks after the second dose of the 4CMenB vaccine was administered, there was no evidence of an hSBA response against the outbreak strain in 33.9% of vaccinees, although no cases of meningococcal disease caused by N. meningitidis B were reported among vaccinated students. (Funded by Princeton University and others.)
Source: N Engl J Med 2016; 375:220-228.
http://www.nejm.org/doi/full/10.1056/NEJMoa1514866
Hemophilus influenzae meningitis and septicaemia in a 14-month-old child after primary immunisation
We report a 14-month-male child, who developed Hemophilus influenzae meningitis after three primary doses of the vaccine. The child presented with fever and seizures. H. influenzae was isolated from both cerebrospinal fluid (CSF) and blood. The child also had features of septicaemia. Procalcitonin (104 ng/ml) and C-reactive protein (CRP – 42.6 mg/dl) were high. Appropriate antibiotics were given. The child made an uneventful recovery. This case highlights vaccine failure, especially after primary immunisation alone.
Source: Indian Journal of Medical Microbiology, Year : 2015 | Volume : 33 | Issue : 1 | Page : 158-160, DOI: 10.4103/0255-0857.148431.
[An infant with meningitis caused by resistant pneumococcus: infection despite vaccination
Abstract
BACKGROUND:
Following the introduction of a heptavalent pneumococcal conjugate vaccine (PCV7) in the Netherlands in 2006, the incidence of invasive pneumococcal disease (IPD) declined significantly. Since then a shift towards non-vaccine serotype IPD has been noted.
CASE DESCRIPTION:
We present the case of multidrug resistant non-vaccine serotype 19A pneumococcal meningitis in a 5-month-old boy. He was admitted to our Paediatric Intensive Care Unit (PICU) with seizures and septic shock. A barbiturate-induced coma was eventually required to control the seizures; shock was combated with intravenous fluids and inotropes. He received a 6-week course of ceftriaxone and vancomycin. At follow-up, one year after discharge, he had unilateral deafness and minor developmental delay.
CONCLUSION:
Worldwide, pneumococcal serotype 19A is now the most common cause of IPD in children, with an increasing incidence of multidrug resistant strains. This trend has not yet been observed in the Netherlands. This case demonstrates that even following the introduction of PCV7 pneumococcal meningitis can still occur. Prompt recognition of the symptoms is still essential.
Source:Ned Tijdschr Geneeskd. 2012;156(1):A3806.
http://www.ncbi.nlm.nih.gov/pubmed/22217305

1 Comment
hi!,I like your writing so much! proportion we keep up a correspondence more approximately your post on AOL? I need an expert in this house to solve my problem. May be that is you! Taking a look forward to look you.